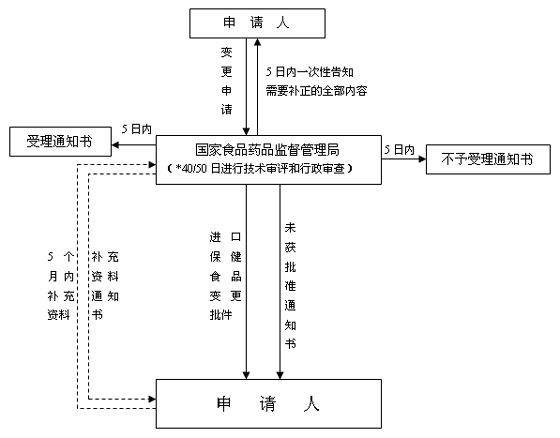
对改变产品名称、保质期、食用量,缩小适宜人群范围,扩大不适宜人群范围、注意事项以及功能项目的变更申请(进口)流程
时间:2022-02-19 浏览:1,254
导读:
* 需要补充资料的注册申请,其审查时限在原审查时限的基础上延长10日。
延长审查时限,从收到最后一次补充资料之日起开始计算。
|
|||
责任编辑:本站小编
相关文章:
- [流程周期]进口保健食品备案申请条件及申报流程
- [流程周期]保健食品注册申请条件及申报流程
- [流程周期]保健食品注册的法律界定、申报流程及资料要求
- [流程周期]保健食品备案信息填报流程图及备案资料形式要求
- [流程周期]天健华成小课堂:保健食品备案流程一览
- [流程周期]对改变产品规格、质量标准以及进口保健食品生产厂商在中国境外改变生产场地的变更申请(进口)
- [流程周期]进口保健食品在境内转让流程图
- [流程周期]进口保健食品在境外转让流程图
- [流程周期]保健食品审批流程图
- [流程周期]功能性保健食品如何快速取得注册批文?
相关推荐:
- [申报资讯]重磅!进口非特殊化妆品纸质批件取消,统一全国电子备案凭证
- [审批动态]2022年07月19日化妆品不予批准决定书待领取信息发布
- [常见问题]化妆品备案注册时多个进口产品列在了同一生产销售证明上,每个产品都需提交销售证明原件吗?如果提交了复印件还需要公证吗?
- [审批动态]2024年08月06日化妆品审评决定书送达信息
- [审批动态]2022年2月28日保健食品批件(决定书)待领取信息-1
- [审批动态]2023年01月05日化妆品决定书送达信息发布
- [常见问题]广东境内牙膏备案及简化备案适用的问题答疑|广州市监局普通化妆品备案问答(四十八期)
- [申报资讯]解读常用保健食品之天然VE概述篇
- [ABOUT]Services
- [常见问题]保健食品申报时生产工艺验证报告和自检报告包括哪些内容?
保健食品申报频道
联系我们
-
86-010-84828041/42
400-6167-168
zhuceabc@zhuceabc.com
咨询微信:
13601366497(化妆品类)
1801335159(特殊食品类)
最新更新
热门排行