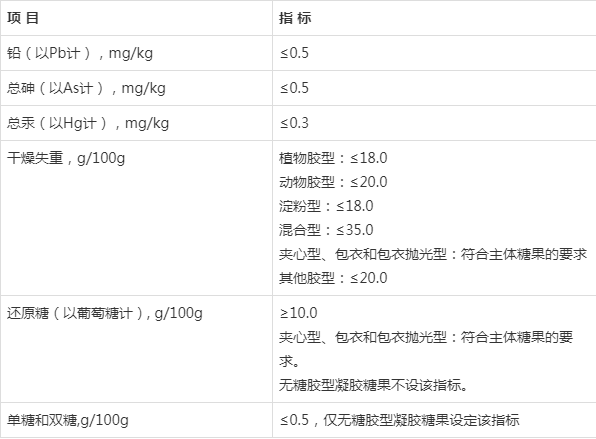
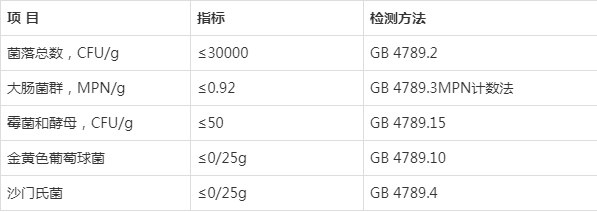
保健食品备案剂型凝胶糖果的技术要求(2020年版)(征求意见稿)及其起草说明
|
|||
相关文章:
- [政策法规]《食品标识监督管理办法》(国家市场监督管理总局规章)
- [政策法规]《 允许保健食品声称的保健功能目录 非营养素补充剂(2023年版)》及配套文件解读
- [政策法规]《保健食品备案产品可用辅料名单及其使用规定(2024年版)》(征求意见稿)起草说明
- [政策法规]保健食品备案产品剂型及技术要求(2024年版)(征求意见稿)起草说明
- [政策法规]保健食品备案产品可用辅料及其使用规定(征求意见稿)(2024年版)
- [政策法规]保健食品备案产品剂型及技术要求(2024年版)(征求意见稿)
- [政策法规]市场监管总局关于发布《保健食品原料人参 西洋参 灵芝备案产品技术要求》的公告
- [政策法规]市场监管总局|关于简化进口保健食品注册备案申请有关领事认证材料的公告
- [政策法规]国家市场监管总局发布 《保健食品标志规范标注指南》
- [政策法规]特殊医学用途配方食品注册管理办法(修订版)
相关推荐:
- [审批动态]2023年05月26日化妆品批准证明文件送达信息发布
- [常见问题]关于化妆品备案注册申报中标签包装的常见问题
- [审批动态]2024年09月27日化妆品注册批准证明文件送达信息
- [常见问题]申报以核酸为原料的保健食品的要求?
- [政策法规]保健食品申报与审评补充规定(试行)
- [常见问题]保健食品注册申请中产品技术要求常见问题及注意事项
- [常见问题]化妆品配方中存在含量不超过0.1%(w/w)的成分,如何标注?
- [审批动态]2023年03月10日化妆品批准证明文件送达信息发布
- [常见问题]【答疑解惑】进口普通化妆品备案资料整理《共性问题错题本》(三)
- [News]NMPA Announcement on Issuing the Rules for Registration and Notification Dossiers of Cosmetics
-
86-010-84828041/42
400-6167-168
zhuceabc@zhuceabc.com
咨询微信:
13601366497(化妆品类)
1801335159(特殊食品类)
- Main Responsibilities of the National Medical Products Administration
- NMPA Announcement on Issuance and Implementation of the Measures for the Administration of Cosmetics Labels
- NMPA Announcement on Updating the Catalogue of Raw Materials Banned for Cosmetics
- NMPA Announcement on Issuing the Catalogue of Used Cosmetic Raw Materials (Edition 2021)
- NMPA Announcement on Issuing the Working Procedures for the Administration of Supplementary Test Methods of Cosmetics and the Technical Guidelines for the Study and Drafting of Supplementary Test Methods of Cosmetics
- Announcement on Issuing the Technical Guidelines for Submitting Registration and Notification Dossier of Cosmetics (Interim)
- NMPA Announcement on Issuing the Classification Rules and Classification Catalogue of Cosmetics
- NMPA Announcement on Issuing the Technical Guidance for the Safety Evaluation of Cosmetics (2021 Edition)
- Contact Us
- Declarations Registered Raiders of Cosmetics
- BeijingTianjianhuacheng International Investment Co.,Ltd
- Guideline of application and evaluation of children’s cosmetics
- Cosmetics declare FAQ
- Procedure
- NMPA Announcement on Updating the Catalogue of Raw Materials Banned for Cosmetics
- Cooperation cases
- NMPA Announcement on Issuing the Technical Guidance for the Safety Evaluation of Cosmetics (2021 Edition)
- EVENTS